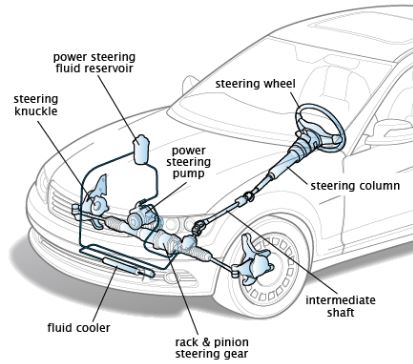
Thermodynamic Equilibrium:
It is said to be thermodynamic Equilibrium when the microscopic properties of the system unchanged and then it is isolated from the surroundings. by the Isolated system in the course of time and which does not gets depart from it, the state of thermodynamic equilibrium is formed.
On the process of this stage, the system does not changes its property.
With the help of three equilibrium stages, the stage of thermodynamic equilibrium is achieved.
- Mechanical
- Chemical.
- Thermal.
1.Mechanical equilibrium:
If there is the balance between all the forces in the system and between the surrounding of the system then it is called as
Mechanical equilibrium this type of system is possible when there is equal pressure in the system and surrounding.
2.Chemical equilibrium:
If the system does not has any chemical reaction and the transfer of matter occurs then it is called as chemical equilibrium.
3.Thermal equilibrium:
If the system has the uniform temperature in the system and in between the system then it is said to be thermal equilibrium.
Please Subscribe! and Don’t forget to Follow us on Facebook, Twitter, Linkedin, Instagram and Google Plus.