Explain Clausius Statement
The Clausius Statement is one of the formulations of the second law of thermodynamics, providing insights into the directionality of heat transfer in natural processes. This statement is named after the German physicist Rudolf Clausius, who played a significant role in developing the foundations of thermodynamics. The Clausius Statement can be succinctly expressed as follows:
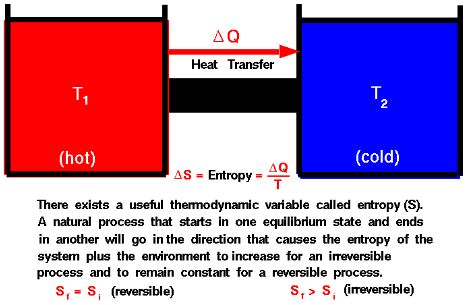
“No process is possible whose sole result is the transfer of heat from a cooler to a hotter body.”
Let’s break down and explain the key elements of the Clausius Statement:
- Heat Transfer Direction:
- The Clausius Statement addresses the spontaneous direction of heat transfer between bodies. It asserts that, under certain conditions, heat energy tends to flow naturally from a hotter object to a colder object. The reverse, where heat transfers spontaneously from a colder object to a hotter one, is deemed impossible according to this statement.
- Reversibility and Irreversibility:
- The statement implicitly considers the reversibility or irreversibility of processes. In a reversible process, the system and its surroundings can be returned to their initial states without a net increase in entropy. In contrast, irreversible processes result in an increase in total entropy, aligning with the second law of thermodynamics.
- Entropic Considerations:
- The concept of entropy plays a crucial role in understanding the Clausius Statement. Entropy (�S) is a measure of the disorder or randomness of a system. For a spontaneous process, the total entropy of an isolated system tends to increase over time.
- Violation Implications:
- If heat were to spontaneously transfer from a colder body to a hotter body without any other effects, it would lead to a decrease in the entropy of the colder body and an increase in the entropy of the hotter body. Such a process would violate the tendency of the total entropy to increase, contradicting the second law of thermodynamics.
- Practical Significance:
- The Clausius Statement has practical significance in various fields, influencing the design and analysis of systems involving heat transfer, such as refrigeration, heat engines, and other thermal processes. It guides engineers and scientists in understanding the limitations and natural tendencies of energy transformations.
In summary, the Clausius Statement provides a fundamental understanding of the irreversibility of certain thermodynamic processes and the inherent directionality of heat transfer. It is a key concept in thermodynamics, contributing to the foundation of our understanding of energy transformations and the behavior of physical systems.
Clausius Statement Proof
The Clausius Statement, which asserts that “no process is possible whose sole result is the transfer of heat from a cooler to a hotter body,” is essentially based on the concept of entropy. To understand the proof of the Clausius Statement, let’s delve into the relationship between heat transfer and entropy change.
Entropy (S) and Heat Transfer: Entropy is a measure of the disorder or randomness of a system. For a reversible process, the change in entropy (ΔS) is related to the heat transfer (Q) by the equation:
ΔS=Q/T
where T is the temperature of the reservoir in kelvins.
Proof of Clausius Statement:
1.Entropy Change for a Reversible Process:
For a reversible process, the entropy change can be expressed as ΔS=Q/T. In this case, the heat transfer is in the same direction as the temperature gradient.
2.Entropy Change for an Irreversible Process:
Now, consider an irreversible process where heat is transferred from a colder body (Tc) to a hotter body (Th). In this case, ΔSirr=Qirr/Th, where Qirr is the heat transferred irreversibly.
3.Total Entropy Change:
The total entropy change for the system and its surroundings is given by ΔStotal=ΔSirr+ΔSres, where ΔSres is the entropy change of the surroundings.
4.Irreversibility and Second Law:
According to the second law of thermodynamics, ΔStotal≥0 for any process. Therefore, ΔSirr+ΔSres≥0.
5.Entropy Change of the Surroundings:
In the case of heat transfer from a colder body to a hotter body, the surroundings (Tres) experience a decrease in entropy (ΔSres<0), compensating for the positive ΔSirr to satisfy the second law.
Conclusion:
The irreversibility of the process requires a decrease in entropy in the surroundings, making the total entropy change nonnegative. This implies that the heat transfer from a colder body to a hotter body is not a spontaneous process, validating the Clausius Statement.
Clausius Statement Example
Let’s consider an example to illustrate the Clausius Statement, emphasizing the directionality of heat transfer in natural processes.
Example: Heat Transfer in a Coffee Cup
Imagine a cup of hot coffee placed on a table in a room. The coffee is initially much hotter than the surrounding air.
According to the Clausius Statement, heat naturally tends to flow from a hotter body to a colder body. In this case:
- Initial State:
- The coffee is the hotter body (higher temperature).
- The surrounding air is the colder body (lower temperature).
- Spontaneous Process:
- Over time, the heat from the hot coffee spontaneously transfers to the colder air in the room.
- Final State:
- Eventually, the coffee cools down, and the air around the coffee warms up slightly.
- Entropy Consideration:
- During this process, the total entropy of the system (coffee and air) increases. The initially ordered state of the hot coffee becomes less ordered as it cools, and the surrounding air gains some thermal disorder.
- Clausius Statement Validation:
- The heat transfer from the hot coffee to the cooler air aligns with the Clausius Statement, which states that no process is possible whose sole result is the transfer of heat from a cooler to a hotter body. In this example, heat spontaneously flows from the hotter coffee to the colder air.
This example illustrates the natural tendency of heat to flow from higher temperatures to lower temperatures, supporting the Clausius Statement. The statement helps explain why a hot cup of coffee cools down over time when exposed to room temperature air, but the reverse process (the coffee spontaneously getting hotter without external influence) is not observed in everyday experiences.
History of Clausius Statement
The Clausius Statement is named after Rudolf Clausius, a German physicist who made significant contributions to the field of thermodynamics during the mid-19th century. Rudolf Clausius formulated this statement as part of his work on the second law of thermodynamics, which describes the directionality of natural processes and the concept of entropy.
Here’s a brief historical overview of Rudolf Clausius and the development of the Clausius Statement:
- Rudolf Clausius (1822-1888):
- Rudolf Clausius was a German mathematician and physicist born on January 2, 1822, in Köslin, Prussia (now Koszalin, Poland). He is considered one of the founders of thermodynamics.
- Contributions to Thermodynamics:
- Clausius is best known for introducing the concept of entropy and formulating the second law of thermodynamics. He made several important contributions to the understanding of heat, work, and energy transformations.
- Introduction of Entropy:
- In the early 1850s, Clausius introduced the concept of entropy as a measure of the amount of thermal energy not available for doing work. He developed the mathematical formulation of entropy and its relationship with heat transfer.
- Development of Second Law:
- Building on the work of Sadi Carnot and others, Clausius formulated the second law of thermodynamics, which describes the limitations and directionality of energy transformations. The formulation of the second law included the famous Clausius Inequality.
- Clausius Statement:
- The Clausius Statement is one of the expressions of the second law and is attributed to Clausius. It emphasizes the impossibility of a process whose sole result is the transfer of heat from a cooler to a hotter body.
- Publication of Works:
- Clausius published his foundational works on thermodynamics in the 1850s and 1860s. One of his notable works is the 1854 paper “On the Motive Power of Heat.”
- Legacy and Recognition:
- Clausius’s contributions to thermodynamics laid the groundwork for the development of the field. His formulation of the second law, including the Clausius Statement, has had a lasting impact on the understanding of energy transformations.
- Later Years and Recognition:
- Clausius continued to work in various areas of physics and mathematics throughout his career. He received recognition for his contributions, including the Copley Medal from the Royal Society in 1879.
Rudolf Clausius’s work significantly influenced the development of thermodynamics and paved the way for later advancements in the understanding of energy, entropy, and the laws governing heat transfer. The Clausius Statement remains a fundamental principle in the study of thermodynamics.
Clausius Statement Application
The Clausius Statement, which states that “no process is possible whose sole result is the transfer of heat from a cooler to a hotter body,” has several applications in various fields. Its implications guide the design, analysis, and understanding of processes involving heat transfer.
Here are some practical applications of the Clausius Statement:
- Refrigeration and Air Conditioning Systems:
- In the design and operation of refrigeration and air conditioning systems, the Clausius Statement is applied to ensure that the heat transfer occurs in the natural direction—from a colder space (refrigerated compartment) to a hotter space (surroundings). It guides engineers in optimizing the efficiency of these systems.
- Heat Engines:
- The Clausius Statement influences the design and efficiency considerations of heat engines, such as steam engines, internal combustion engines, and gas turbines. Engineers use the statement to set realistic expectations for the direction of heat flow in these systems.
- Thermal Power Plants:
- In the context of thermal power plants, which convert heat energy into electricity, the Clausius Statement guides the optimization of the overall efficiency of the power generation process. Heat is transferred from a high-temperature source to a lower-temperature sink, consistent with the statement.
- Heat Exchangers:
- Heat exchangers are widely used in industrial processes, HVAC systems, and various applications. The Clausius Statement is considered in the design of heat exchangers to ensure that heat transfer aligns with the natural direction—from higher to lower temperatures.
- Energy Conservation:
- The Clausius Statement plays a role in energy conservation strategies. Understanding the limitations imposed by the statement helps policymakers and engineers develop energy-efficient technologies and practices, promoting sustainable energy use.
- Thermoelectric Devices:
- Devices that convert temperature differences into electrical energy (thermoelectric devices) adhere to the principles outlined in the Clausius Statement. These devices are used in applications such as waste heat recovery.
- Material Science and Heat Treatment:
- The Clausius Statement is considered in material science, especially in processes involving heat treatment. Heat transfer during annealing, quenching, and tempering processes is designed to align with the natural direction of heat flow.
- Environmental Impact Assessments:
- In environmental studies and assessments, the Clausius Statement is a fundamental principle when evaluating the impact of heat release or absorption in various processes. It contributes to a comprehensive understanding of the thermodynamic aspects of environmental systems.
- Educational Tools:
- The Clausius Statement is an integral part of thermodynamics education. It serves as a foundational concept for students studying heat transfer, energy conversion, and the second law of thermodynamics.
- Policy and Regulations:
- Governments and regulatory bodies use the principles of the Clausius Statement when formulating regulations related to energy efficiency, waste heat recovery, and environmental sustainability.
Frequently Asked Questions
1.What is the Clausius Statement in thermodynamics?
The Clausius Statement asserts that no process is possible whose sole result is the transfer of heat from a cooler to a hotter body.
2.How does the Clausius Statement relate to the direction of heat transfer?
The statement emphasizes that heat tends to flow spontaneously from hotter to colder bodies and prohibits the natural transfer of heat from a cooler to a hotter body.
3.Is the Clausius Statement a fundamental principle in thermodynamics?
Yes, the Clausius Statement is one of the expressions of the second law of thermodynamics, providing insights into the irreversibility and directionality of natural processes.
4.Can you provide a real-world example illustrating the Clausius Statement?
A: Certainly! When a hot cup of coffee cools down in a room, heat naturally flows from the hotter coffee to the cooler room air, aligning with the Clausius Statement.
5.How does the Clausius Statement impact the design of refrigeration systems?
In refrigeration systems, the statement guides engineers to ensure that heat is transferred from the refrigerated space (cooler) to the surroundings (warmer), adhering to the natural direction of heat flow.
6.Does the Clausius Statement apply to all heat transfer processes?
Yes, the statement is a universal principle and applies to all processes involving heat transfer. It sets a fundamental constraint on the directionality of heat flow.
7.Can the Clausius Statement be violated in any practical scenario?
No, the Clausius Statement is a fundamental principle in thermodynamics, and there is no known practical scenario where it can be violated. It aligns with our everyday observations of heat transfer.
8.How does the Clausius Statement contribute to energy efficiency considerations?
The statement guides the design of energy-efficient systems by ensuring that heat is utilized in a way that aligns with the natural direction of heat transfer, minimizing wasteful processes.
9.What is the relationship between the Clausius Statement and the concept of entropy?
The Clausius Statement is closely related to the increase in entropy, as heat transfer from hotter to colder bodies leads to an increase in the overall entropy of the system, in accordance with the second law of thermodynamics.
10.How is the Clausius Statement taught in thermodynamics education?
In thermodynamics courses, the Clausius Statement is introduced as a key principle in understanding the irreversibility of processes and the limitations on heat transfer, providing a foundation for more advanced concepts.