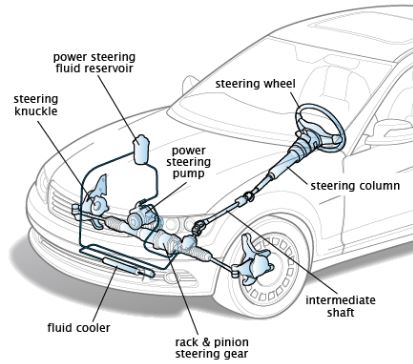
Charles Law:
Charles Law states that for a given mass of gas at constant pressure, the volume V is directly proportional to its thermo-dynamic temperature T
i.e. V ∝ T
or V = kT
or
V/T = k at constant pressure, where
T = thermodynamic temperature in
Kelvin (K).
A process that takes place at constant pressure is called an isobaric process.
The relationship between the Celsius scale of temperature and the thermodynamic or absolute scale is given by:
kelvin = degrees Celsius + 273
i.e. K = °C + 273
or °C = K – 273 (as stated in Chapter 20).
If a given mass of gas at a constant pressure occupies a volume V1 at a temperature T1 and a volume V2 at temperature T2, then
V1/T1= V2/T2