Adiabatic Process:
Adiabatic Process: In this article we will learn about Adiabatic Process. What is Adiabatic Process? How Adiabatic Process works? On which principle Adiabatic Process works and types of adiabatic process. The detailed explanation about Adiabatic Process we will discuss in this article. The adiabatic process works on the main principle of thermodynamic process. Adiabatic process explains clearly about energy transfer to surroundings only by work done on it. There is no direct changes occur in mass, heat transfer etc.Energy transfer takes place by only work done, there is no another heat/matter directly take places in it, this type of system is called as adiabatically isolated.
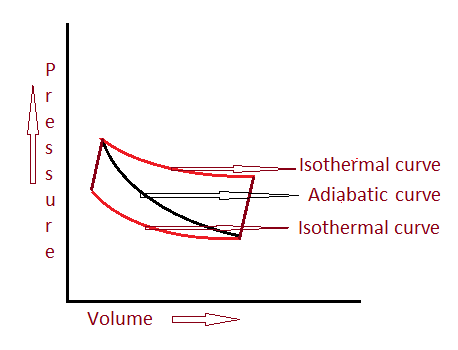
Example of an adiabatic process we can observe around us like when a gas is compressed in a closed area heat gets increased there is no heat/mass transfer take place but work done on it develops to high temperature this also increases the pressure. And generally, it gets cooled when it expanded.
By this, we can observe that the changes occurred by this process is pressure, volume, temperature.
Types of Adiabatic Process:
There are two types of Adiabatic Process they are as follows:
1. Reversible Adiabatic Process
2. Irreversible Adiabatic Process
Reversible Adiabatic Process:
In Reversible Adiabatic Process entropy remains constant
Irreversible Adiabatic Process:
In Irreversible Adiabatic Process entropy will not remain constant